Digital Health Technology Used in Emergency Large-Scale Vaccination Campaigns in Low- and Middle-Income Countries: A Narrative Review for Improved Pandemic Preparedness
Janssen Pharmaceutica NV (Kenna, Willems, Masynm, Pattery); Akkodis Belgium (Broadfield); Janssen Research & Development (Draghia-Akli)
"Through the sharing lessons learned in various LMICs with different tools, the global community can collectively gain knowledge to be best prepared for a future pandemic response."
Evidence suggests that large-scale vaccination campaigns can benefit from using digital health tools, particularly in low- and middle-income countries (LMICs). This narrative review examines the functionalities and technical specifications of these tools with the hope of providing a resource for LMICs to draw on in future large-scale vaccination campaigns for outbreak response based on experiences in similar settings. The paper also considers topics such as data security and safety, tool availability, costs, challenges specific to LMICs, and options in ensuring sustainability of tools.
In October-November 2022, the researchers conducted a narrative review in PubMed and the grey literature for data available within 5 years; they performed additional searches in PubMed in January 2023. Out of the 18 tools identified through the review, 9 were developed specifically for COVID-19, and others have been used in previous large-scale immunisation campaigns in emergency settings. For example, the Oracle Health Management System was used in a yellow fever vaccination campaign in Ghana, and Vxnaid was used in a large-scale, multi-dose, Ebola vaccination campaign in Rwanda. The functionalities of the digital health tools that were identified are summarised in Table 1 of the paper and their technical specifications outlined in Table 2. Additional details about tool functionalities can be found in Supplemental Table S1.
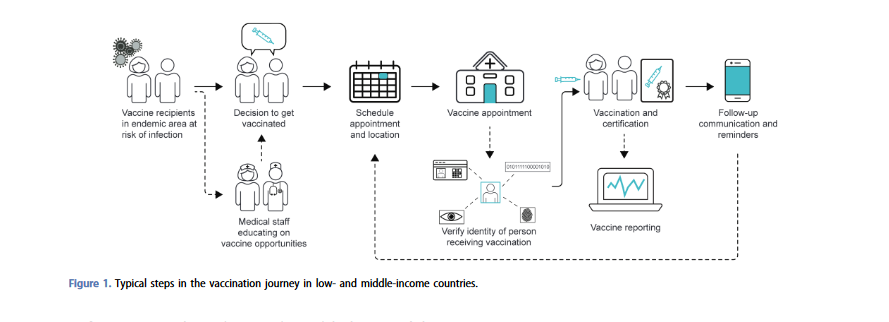
There are well-defined steps for a typical vaccination process at a healthcare centre facility delivering a large-scale vaccination campaign in response to outbreaks, which are mostly consistent across countries. The narrative review focuses on the use of digital health tools along these steps. For example:
- Appointment scheduling: Most tools need internet connectivity for scheduling an appointment online. mVacciNation offers self-registration for an appointment via unstructured supplementary service data (USSD), short messaging service (SMS), WhatsApp, or web application. Sometimes additional features are incorporated together with appointment scheduling. For example, India's Co-WIN system allows users to select convenient vaccine clinic locations and vaccine type or brand. Many tools that offer appointment scheduling also generate appointment confirmations or reminders, and some may track and follow-up defaulters who have missed a vaccination appointment (e.g., mVacciNation).
- Personal identification: Many people worldwide, especially those living in LMICs, lack an official proof of identity. Based upon the learnings from recent Ebola outbreaks, it is important that biometric tools are contactless, like iris scanning or facial recognition. If biometric identification is not possible, alternative identification modalities can include the manual entry of a unique identifier, QR/barcode scanning of a person's campaign card, or voucher supported by some tools. The Oracle Health Management System creates an electronic health record in a cloud database for every person vaccinated, highlighting the usefulness of a cloud-based tool.
- Vaccine verification: Few of the digital health tools identified in the review are able to address vaccine verification. For example, Vxnaid can scan a vaccine vial barcode to document the manufacturer and lot number with a built-in warning system if a healthcare worker accidentally selects an unscheduled vaccine for use.
- Vaccine certification: Typically, vaccine records and certifications that stay with the individual have been paper-based, but digital tools offer a wide range of features, including: storing vaccine records on- and offline; generating, signing, and verifying vaccine certificates from a health authority; and linking certificates to individuals via secure, confidential methods.
- Vaccine reporting: Digital health tools can ease the work of healthcare workers or data entry clerks by making vaccination status and numbers of individuals vaccinated directly available via dashboard reports. Tools as DHIS2 COVID-19, OpenSRP FHIR Core, and Trusted Vaccines can collect and track individual immunisation data in national electronic immunisation registries. Additionally, some tools provide data visualisations, allowing campaign management and field teams to make timely data-driven decisions to meet coverage targets and optimise operations - for example, Vxnaid, Co-WIN, DHIS2 COVID-19, and CommCare.
- Follow-up communication and appointment reminders for multi-dose vaccine regimens or booster doses: Communication with vaccinated individuals keeps them informed and up to date and may improve adherence to their immunisation schedule. For example, Vxnaid can utilise SMS, WhatsApp messages, or pre-recorded voicemail messages in any language, and additional content can also be created and shared with users to address health and vaccine misinformation, provide updates on travel restrictions, and inform users on practical changes to vaccine appointments. The WelTel Health Platform can broadcast informational health videos; its reminders serve as a call-to-action, and the tool allows real-time communication between vaccine recipients and healthcare providers.
As noted here, for LMICs looking to integrate and implement new digital health tools into their existing systems, it is important take into account the appropriateness of the tool in addressing their need(s), and the associated costs. Digital Public Good (DPG) labeled tools may be good options to consider. The DPG standard, maintained by the DPG Alliance, assesses tools across multiple factors, including being open-source.
Also, data privacy and security measures are essential for the ethical use of digital health technology. The World Health Organization (WHO) Global Strategy on Digital Health, the Principles for Digital Development, the DPG standard from the DPG Alliance, and the Digital Global Good (DGG) standard maintained by Digital Square all emphasise the need to be transparent about how data will be collected and used, to plan for and thwart security breaches, and to protect data against harmful or inappropriate use. These guidelines recommend addressing the protection of privacy and data security of digital tools already at the design stage. To protect data privacy and ensure data security, countries need to create and implement a robust legal and regulatory framework at national and international levels around the use of personal data with clearly defined roles for data governance for equitable data sharing, which does not yet exist in all LMICs.
The paper goes on to look at challenges and opportunities for implementing digital health tools in LMICs. Selected highlights:
- Developing tools with a human-centred design approach allows for local stakeholder input and can develop fit-for-purpose tools that support the local vaccination process and understand interdependencies with other systems.
- It is critical that the digital tools themselves do not become bottlenecks to efficient vaccination strategies. High-quality data can be used for informed decision making on bottlenecks.
- Acceptance of biometric identification tools may be perceived to present challenges, with the idea that local beliefs and (mis)conceptions may erode trust in certain digital identification methods. It is important to determine the ideal option for a given setting and community and possibly to ensure that alternative accepted identification options are available to support the success of a large-scale vaccination campaign.
- Although digital health technologies provide opportunities to support inclusiveness and promote health equity, their adoption can be hampered by gender-based, linguistic, and digital literacy barriers. Governments should, for example, considering using tools that support multiple communication methods. Vxnaid offers both text messages and automated voice messages for reminders and for communicating key information to vaccine recipients, and Co-WIN offers an interactive voice response system (IVRS) that allows the registration of multiple individuals to a single phone number, helping to reach individuals without smartphones or sufficient digital literacy. The support from telecommunication companies to provide global coverage at affordable rates, even in remote areas, will allow for increased adoption of digital health tools and access to more vulnerable communities with previously limited access to health care.
- Improving internet connectivity in LMICs will also facilitate adoption.
Next, the researchers explore considerations for forward-thinking and vaccine campaign sustainability. Echoing a recommendation made earlier in the paper, they urge that, when starting from an empty digital space, the probability of being sustainable can be heightened by co-designing innovative digital technologies together with different stakeholders who will be involved in the project implementation, including end users, government and healthcare stakeholders, and telecommunication companies. In addition to verifying open-source status, the DPG or DGG standards noted above also assess maturity, quality, and sustainability aspects of a digital tool and can be useful when reviewing various tool options.
In conclusion, though digital tools will not solve all of the challenges faced by LMICs in their efforts to set up large-scale vaccination campaigns, such as vaccine hesitancy, the researchers suggest that the adoption of digital technology can improve equity for people living in LMICs in a global pandemic situation. To that end, this review may aid LMICs still needing to prepare large-scale vaccination campaigns in the selection of supporting digital health tools. "Further research on impact and cost-effectiveness is needed."
Expert Review of Vaccines 2023, Vol. 22, No. 1, 243-55. https://doi.org/10.1080/14760584.2023.2184091.
- Log in to post comments












































